Recursion Provides Business Updates and Reports Third Quarter 2021 Financials
- Received Fast Track Designation for REC-2282, a potential treatment for NF2 meningiomas, and expect to enroll the first patient in a Phase 2/3 trial in early 2022
- Advanced REC-994, a potential treatment for CCM, and now expect to enroll the first patient in a phase 2 trial in early 2022
- Received Orphan Drug Designation from the FDA for REC-4881, a potential treatment for FAP, and expect to enroll the first patient in a Phase 2 trial in the first half of 2022
- Continued to advance multiple simultaneous discovery programs in fibrosis with Bayer
- Accelerated investment in the industrialization of chemistry to complement its longstanding work to decode biology
SALT LAKE CITY, Nov. 10, 2021 /PRNewswire/ -- Recursion (Nasdaq : RXRX), a clinical-stage biotechnology company decoding biology by integrating technological innovations across biology, chemistry, automation, data science and engineering, today reported business updates and financial results for its third quarter ending September 30, 2021.
"In Q3, our team made progress towards our vision to industrialize drug discovery. We are now harvesting the efforts of the past few years to build a map of human cellular biology through the continued refinement and increased usage of our inference-based approach to drug discovery. With the power of our Recursion Map illuminating new and exciting relationships in biology, we are now deeply focused on extending our chemistry capabilities to significantly improve, scale and speed up new chemical entity development to address the plethora of novel biological relationships we are discovering," said Recursion Co-Founder & CEO Chris Gibson, Ph.D. "In addition, our rapidly growing development team is preparing for our four clinical-stage programs to initiate Phase 2 or Phase 2/3 studies in the first half of 2022, including two of the programs that we expect will enroll their first patients in early 2022. To facilitate our broad ambition, we continue to rapidly grow our workforce while nurturing Recursion's culture and community."
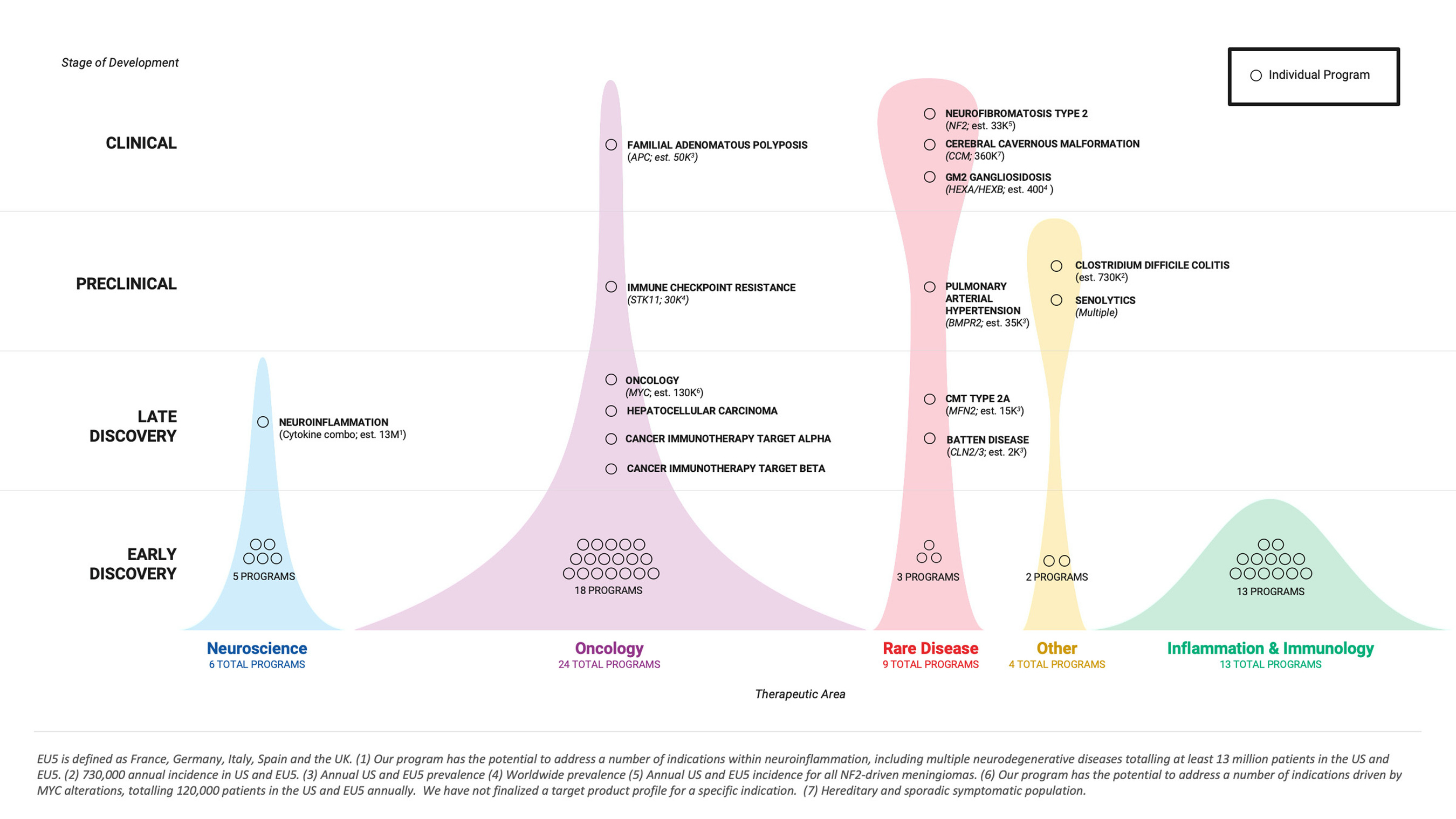
Recursion finished the third quarter of 2021 with a portfolio of 4 clinical stage programs, 4 preclinical programs, 7 late discovery programs, and 41 early discovery programs. Additionally, Recursion continued scaling the total number of executed phenomic experiments to approximately 95 million, the size of its proprietary data universe to over 11 petabytes, and the number of biological inferences to approximately 200 billion. Data have been generated on the Recursion OS across 38 human cell types, an in-house chemical library of over 717 thousand compounds, and an in silico library of 12 billion small molecules, by a growing team of more than 330 Recursionauts that is balanced between life scientists and computational and technical experts.
Summary of Business Highlights
- Clinical Programs
- Neurofibromatosis type 2 (NF2) (REC-2282): In early October we received Fast Track Designation for REC-2282 from the FDA for the potential treatment of NF2 meningiomas. We plan to initiate a parallel group, two stage, Phase 2/3, randomized, multicenter study in early 2022.
- Cerebral cavernous malformation (CCM) (REC-994): We plan to initiate a Phase 2, double-blind, placebo-controlled safety, tolerability and exploratory efficacy study of this candidate in early 2022.
- Familial adenomatous polyposis (FAP) (REC-4881): In September we received Orphan Drug Designation for REC-4881 from the FDA for the potential treatment of Familial Adenomatous Polyposis. We plan to initiate a Phase 2, randomized, double-blind, placebo-controlled study to evaluate safety, pharmacokinetics and efficacy in the first half of 2022.
- GM2 gangliosidosis (REC-3599): We plan to initiate a Phase 2 study of this candidate in the first half of 2022.
- Preclinical Programs
- Clostridium difficile colitis (REC-3964): We expanded our medicinal chemistry team and digital chemistry tools and made progress in IND-enabling studies for REC-3964, which is the most advanced New Chemical Entity developed by the Recursion OS.
- Bayer AG Partnership: We continue to advance our collaboration with Bayer to discover small molecule drug candidates with the potential to treat fibrotic diseases. We have multiple programs progressing simultaneously with our partner.
- Recursion OS
- Biological Contexts: We advanced our capabilities to model diseases in multiple biological contexts, including new types of biological perturbations beyond CRISPR-based knockouts, complex cell type onboarding, and organoid model systems. Moreover, we made progress on multiple maps in iPSC-derived neural cell types.
- Mechanisms of Action: We improved our computational methods to identify mechanisms of action and used this technology to increase our ability to screen out compounds with potentially toxic effects for multiple programs earlier than is possible with traditional approaches. We believe that such methods will better enable us to advance the most promising novel chemical compounds through discovery.
- Transcriptomics Validation: We made significant improvements to our transcriptomics protocols to enable increases in throughput. Additionally, we have been optimizing our ability to use transcriptomics signatures for compound characterization.
- Facilities and Manufacturing: We continued to make progress in expanding our current headquarters and creating a chemistry, manufacturing and controls (CMC) site in Salt Lake City. These spaces are designed with flexibility in mind to enable next-generation automated workflows and instruments for compound, tissue culture, and biobank management to further industrialize the drug discovery and development process.
Third Quarter 2021 Financial Results
- Cash Position: Cash, cash equivalents, and investments were $578.9 million as of September 30, 2021.
- Revenue: Total revenue, consisting primarily of revenue from collaborative agreements, was $2.5 million for the third quarter of 2021, compared to $1.0 million for the third quarter of 2020. The increase was due to revenue recognized from our collaboration with Bayer.
- Research and Development Expenses: Research and development expenses were $33.2 million for the third quarter of 2021, compared to $16.5 million for the third quarter of 2020. The increase in research and development expenses was primarily due to an increased number of experiments run on the Recursion OS, an increased number of assets being validated, and increased clinical costs as studies progressed.
- General and Administrative Expenses: General and administrative expenses were $15.7 million for the third quarter of 2021, compared to $7.0 million for the third quarter of 2020. The increase in general and administrative expenses was due to the growth in size of the company's operations, including an increase in salaries and wages of $3.7 million, equipment costs, human resources-related costs, facilities costs, and other administrative costs associated with operating a high-growth company.
- Net Loss: Net loss was $47.4 million for the third quarter of 2021, compared to a net loss of $23.9 million for the third quarter of 2020.
Additional Corporate Updates
- Operations in Canada: Jordan Christensen joined Recursion as Vice President, Engineering and also became our Toronto Site Lead. Additionally, we opened our Montreal office and hired multiple machine learning research scientists.
- Translational Biology: Alison O'Mahony, Ph.D., joined Recursion as Vice President, Discovery Platform and will be responsible for continued scaling and improvement of Recursion's orthogonal validation and bespoke validation assays to continue driving down the time from initial discovery to clinical development. Dr. O'Mahony previously served as Vice President, Translational Biology at Eurofins Discovery.
- Information Security: Ganesh Jagannathan joined Recursion as Chief Information Security Officer & Vice President, Information Technology and will be responsible for all strategic, innovative and operational aspects of Information Security and Information Technology. Mr. Jagannathan previously served as Chief Information Security Officer at Jazz Pharmaceuticals.
- CEO Rule 10b5-1 Plans: Chris Gibson, Ph.D., the company's Co-Founder and CEO, established personal stock trading plans in the second quarter of 2021 in accordance with Rule 10b5-1 under the Securities and Exchange Act of 1934 and Recursion's insider trading policy. Under the plans, all outstanding stock options may be exercised and we anticipate shares representing up to approximately 4% of Dr. Gibson's holdings may be sold or transferred to donor-advised philanthropic funds. We anticipate the Rule 10b5-1 transactions may take place over the next 13 months. Any such transactions will be disclosed through public filings as required by the SEC.
About Recursion
Recursion is a clinical-stage biotechnology company decoding biology by integrating technological innovations across biology, chemistry, automation, machine learning and engineering. Our goal is to radically improve the lives of patients and industrialize drug discovery. Central to our mission is the Recursion Operating System, which combines an advanced infrastructure layer to generate what we believe is one of the world's largest and fastest-growing proprietary biological and chemical datasets. We combine that with the Recursion Map, a suite of custom software, algorithms, and machine learning tools that we use to explore foundational biology unconstrained by human bias and navigate to new biological insights. We are a biotechnology company scaling more like a technology company. Learn more at www.Recursion.com, or connect on Twitter and LinkedIn. Recursion is also a founding member of BioHive, the Utah life sciences industry collective.
Contact
Elyse Freeman - Senior Communications and Content Manager
InvestorRelations@Recursion.com
Condensed Consolidated Statements of Operations
|
Recursion Pharmaceuticals, Inc. Condensed Consolidated Statements of Operations (unaudited) (in thousands, except share and per share amounts) |
||||||
|
Three months ended |
Nine months ended |
|||||
|
September 30, |
September 30, |
|||||
|
Revenue |
2021 |
2020 |
2021 |
2020 |
||
|
Grant revenue |
$ 34 |
$ 163 |
$ 145 |
$ 409 |
||
|
Operating revenue |
2,500 |
862 |
7,500 |
862 |
||
|
Total revenue |
2,534 |
1,025 |
7,645 |
1,271 |
||
|
Operating expenses |
||||||
|
Research and development |
33,246 |
16,535 |
86,979 |
42,621 |
||
|
General and administrative |
15,690 |
6,964 |
38,481 |
17,684 |
||
|
Total operating expenses |
48,936 |
23,499 |
125,460 |
60,305 |
||
|
Loss from operations |
(46,402) |
(22,474) |
(117,815) |
(59,034) |
||
|
Other loss, net |
(1,026) |
(1,399) |
(3,731) |
(2,206) |
||
|
Net loss |
$ (47,428) |
$ (23,873) |
$ (121,546) |
$ (61,240) |
||
|
Per share data |
||||||
|
Net loss per share of Class A and B common stock, basic and diluted |
$ (0.28) |
$ (1.09) |
$ (1.10) |
$ (2.82) |
||
|
Weighted average shares (Class A and B) outstanding, basic and diluted |
168,533,550 |
21,817,900 |
110,513,231 |
21,704,008 |
||
Condensed Consolidated Balance Sheets
|
Recursion Pharmaceuticals, Inc. Condensed Consolidated Balance Sheets (unaudited) (in thousands) |
|||
|
September 30, |
December 31, |
||
|
2021 |
2020 |
||
|
Assets |
|||
|
Current assets |
|||
|
Cash and cash equivalents |
$ 394,721 |
$ 262,126 |
|
|
Restricted cash |
10,233 |
5,041 |
|
|
Accounts receivable |
34 |
156 |
|
|
Other receivables |
2,248 |
- |
|
|
Investments |
184,189 |
- |
|
|
Other current assets |
9,445 |
2,155 |
|
|
Total current assets |
600,870 |
269,478 |
|
|
Property and equipment, net |
55,439 |
25,967 |
|
|
Intangible assets, net |
2,262 |
2,490 |
|
|
Other non-current assets |
35 |
650 |
|
|
Total assets |
$ 658,606 |
$ 298,585 |
|
|
Liabilities, convertible preferred stock and stockholders' equity (deficit) |
|||
|
Current liabilities |
|||
|
Accounts payable |
$ 6,326 |
$ 1,074 |
|
|
Accrued expenses and other liabilities |
25,113 |
10,485 |
|
|
Current portion of unearned revenue |
10,000 |
10,000 |
|
|
Current portion of notes payable |
88 |
1,073 |
|
|
Current portion of lease incentive obligation |
1,416 |
467 |
|
|
Total current liabilities |
42,943 |
23,099 |
|
|
Deferred rent |
3,348 |
2,674 |
|
|
Unearned revenue, net of current portion |
9,167 |
16,667 |
|
|
Notes payable, net of current portion |
656 |
11,414 |
|
|
Lease incentive obligation, net of current portion |
3,460 |
2,708 |
|
|
Total liabilities |
59,574 |
56,562 |
|
|
Commitments and contingencies |
|||
|
Convertible preferred stock |
- |
448,312 |
|
|
Stockholders' equity (deficit) |
|||
|
Common stock (Class A and B) |
2 |
- |
|
|
Additional paid-in capital |
934,175 |
7,312 |
|
|
Accumulated deficit |
(335,147) |
(213,601) |
|
|
Accumulated other comprehensive income |
2 |
- |
|
|
Total stockholder's equity (deficit) |
599,032 |
(206,289) |
|
|
Total liabilities, convertible preferred stock and stockholders' equity (deficit) |
$ 658,606 |
$ 298,585 |
|
Forward-Looking Statements
This document contains information that includes or is based upon "forward-looking statements" within the meaning of the Securities Litigation Reform Act of 1995, including, without limitation, those regarding Recursion's mission; early and late stage discovery, preclinical, and clinical programs; collaborations; prospective products and their future indications and market opportunities; Recursion OS and other technologies; expansion of facilities and expected uses; workforce growth; employee stock trading plans; business and financial performance; and all other statements that are not historical facts. Forward-looking statements may or may not include identifying words such as "plan," "will," "expect," "anticipate," "intend," "believe," "potential," "continue," and similar terms. These statements are subject to known or unknown risks and uncertainties that could cause actual results to differ materially from those expressed or implied in such statements, including but not limited to: challenges inherent in pharmaceutical research and development, including the timing and results of preclinical and clinical programs, where the risk of failure is high and failure can occur at any stage prior to regulatory approval due to lack of sufficient efficacy, safety considerations, or other factors; our ability to leverage and enhance our drug discovery platform; our ability to obtain financing for development activities and other corporate purposes; the success of our collaboration activities; our ability to obtain regulatory approval of, and ultimately commercialize, drug candidates; the impact of the COVID-19 pandemic; our ability to obtain, maintain, and enforce intellectual property protections; cyberattacks or other disruptions to our technology systems; our ability to attract, motivate, and retain key employees and manage our growth; and other risks and uncertainties described under the heading "Risk Factors" in our filings with the U.S. Securities and Exchange Commission, including our most recent Quarterly Report on Form 10-Q. All forward-looking statements are based on management's current estimates, projections, and assumptions, and Recursion undertakes no obligation to correct or update any such statements, whether as a result of new information, future developments, or otherwise, except to the extent required by applicable law.
SOURCE Recursion